I.V. de Jong-Medvetska MD, W.G. Vermeulen-van Eck and R.C.W. Vermeulen PhD
Several studies reported the effect of L-carnitine in CFS/ME and chronic fatigue [1-3]. In these studies the change that was attributed to L-carnitine was measured by global impression of change and by questionnaires. In 2006 we introduced the repeated cardiopulmonary exercise test (CPX) in the CFS Centre and we selected a test for speed and accuracy of response from the Amsterdam Neuropsychological Tasks [4, 5]. Recently we expanded the CPX to calculate the oxygen uptake by muscle cells. This allows us to measure changes in the metabolism of these cells during incremental exercise [6]. In September 2013 we started to use these tests clinically in patients who visited the CFS/ME Medical Centre in Amsterdam for treatment with L-carnitine.
Clinical protocol
Before the first visit patients completed questionnaires that measure severity of fatigue [7], Physical impairment [8], additional symptoms [9], depression [10], pain [11], sleepiness [12], and mental health [13] for the diagnoses of CFS [14], and a questionnaire for the diagnosis of Myalgic Encephalitis (ME) [15].
During the first visit the history was discussed in an interview, followed by a test for speed and accuracy of response. EKG, tilt table test, and measurement of the resting metabolic rate. During the second visit the physical impairment was measured by CPX. During the third visit the speed and accuracy of response test and the CPX were repeated. During the next 2 weeks the patient measured the heart rate at home.
The diagnostic protocol ended with a concluding visit after 2 weeks. During this study L-carnitine was offered to all patients who were eligible. After 2 months the test for speed and accuracy of response and the CPX were repeated and L-carnitine dosage was adapted.
Results
71 patients were eligible for L-carnitine medication. The results of the patients who finished the 2 months of therapy in the period of November 2013 till April 2014 are presented in table 1 and 2.
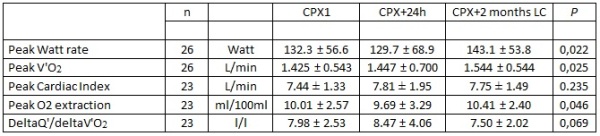
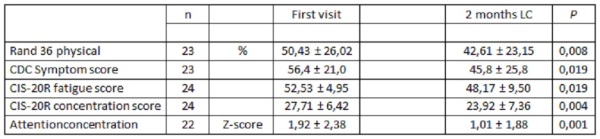
The maximal exercise capacity after 2 months of L-carnitine was higher in 18 patients, lower in 7 patients and unchanged in 1 patient. Oxygen extraction by the muscle cells improved and the compensating increase of the cardiac output relative to the increase of the O2 uptake declined. The self-rated exercise capacity (Rand-36 physical) varied widely and showed a decline, while the CDC and CIS values improved. The test for speed and accuracy of response showed a clear improvement during L-carnitine medication.
Discussion
The 2 objective tests, the cardiopulmonary exercise test and the Shifting Attentional Test, showed that physical and cognitive function improved in this population during L-carnitine treatment. The increase of the peak O2 uptake was explained by the increase of the O2 extraction by the muscle cells during L-carnitine. It is not unlikely that the improvement of cognitive function was also due to the improved production of ATP. The results of the questionnaires improved as well, but the changes of the CDC and CIS-20R were not correlated with the changes of peak VO2 or peak Watt rate. This indicates that the subjective measures may be of little value as marker for clinical change.
The results of this analysis are indicative only because of the uncontrolled collection of data and no proof of a positive effect of L-carnitine in CFS/ME as was reported in our previous study [2].
Conclusion
The positive effect of L-carnitine that was reported in previous studies was confirmed and the results indicate that objective tests for improvement of physical and cognitive function confirm the clinical global impression of change. The change of peak V’O2 was explained by an improved energy production in the muscle cells.
Table 1. Means and Standard Deviations of Peak watt rate, peak O2 uptake, peak cardiac index, peak O2 extraction and increase of cardiac output relative to increase of O2 uptake during incremental exercise in CPX1, CPX 24h after CPX1 and CPX after 2 months L-carnitine medication. P: paired samples T-test CPX1 and CPX during L-carnitine.
Table 2. Mean and Stendard Deviation of Physical axis of Rand36, CDC symptom score-DLV, fatigue and concentration axis of CIS-20R and response time of Shifting Attentional Test Visual of ANT. P: paired samples T-test first visit and during 2 months L-carnitine.
1. Plioplys AV, Plioplys S: Amantadine and L-Carnitine treatment of chronic fatigue syndrome. Neuropsychobiology 1997, 35(1):16-23.
2. Vermeulen RCW, Scholte HR: Exploratory Open Label, Randomized Study of Acetyl- and Propionylcarnitine in Chronic Fatigue Syndrome. Psychosomatic Medicine 2004, 66(2):276-282.
3. Malaguarnera M, Gargante MP, Cristaldi E, Colonna V, Messano M, Koverech A, Neri S, Vacante M, Cammalleri L, Motta M: Acetyl l-carnitine (ALC) treatment in elderly patients with fatigue. Archives of Gerontology and Geriatrics 2008, 46(2):181-190.
4. Vermeulen RCW, Kurk RM, Visser FC, Sluiter W, Scholte HR: Patients with chronic fatigue syndrome performed worse than controls in a controlled repeated exercise study despite a normal oxidative phosphorylation capacity. Journal of Translational Medicine 2010, 8:93.
5. De Sonneville LMJ: Amsterdam Neuropsychological Tasks: a computer-aided assessment program. In: Cognitive ergonomics, clinical assessment and computer-assisted learning: Computers in Psychology. Volume 6, edn. Edited by Den Brinker BPLM, Beek PJ, Brand AN, Maarse SJ, Mulder LJM. Lisse: Swets & Zeitlinger; 1999: 187-203.
6. Vermeulen RCW, Vermeulen-van Eck WG: Decreased oxygen extraction during cardiopulmonary exercise test in patients with chronic fatigue syndrome. Journal of Translational Medicine 2014, 12(20).
7. Vercoulen JHHM, Alberts M, Bleijenberg G: The Checklist Individual Strength (CIS). Gedragstherapie 1999, 32:31-36.
8. Aaronson NK, Muller M, Cohen PDA, Essink-Bot ML, Fekkes M, Sanderman R, Sprangers MAG, te Velde A, Verrips E: Translation, Validation, and Norming of the Dutch Language Version of the SF-36 Health Survey in Community and Chronic Disease Populations. Journal of Clinical Epidemiology 1998, 51(11):1055-1068.
9. Vermeulen RCW: Translation and validation of the Dutch language version of the CDC Symptom Inventory for assessment of Chronic Fatigue Syndrome (CFS). Population Health Metrics 2006, 4(-):12.
10. Zung WW: A self-rating depression scale. Archives of General Psychiatry 1965 12:63 – 70.
11. Van Der Kloot WA, Oostendorp RAB, Van Der Meij J, Van Den Heuvel J: The Dutch version of the McGill pain questionnaire: A reliable instrument. Nederlands Tijdschrift voor Geneeskunde 1995, 139(13):669-673.
12. Johns MW: A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep 1991, 14(6):540- 545.
13. Terluin B, Van Rhenen W, Schaufeli WB, De Haan M: The four-dimensional symptom questionnaire (4DSQ): Measuring distress and other mental health problems in a working population. Work and Stress 2004, 18(3):187-207.
14. Fukuda K, Straus SE, Hickie I, Sharpe MC, Dobbins JG, Komaroff A, Schluederberg A, Jones JF, Lloyd AR, Wessely S et al: The chronic fatigue syndrome: A comprehensive approach to its definition and study. Annals of Internal Medicine 1994, 121(12):953-959.
15. Carruthers BM, Van de Sande MI, De Meirleir KL, Klimas NG, Broderick G, Mitchell T, Staines D, Powles ACP, Speight N, Vallings R et al: Myalgic encephalomyelitis: International Consensus Criteria. Journal of Internal Medicine 2011, 270(4):327-338.